When they’re not busy identifying bees from our research fields, the pollinator teaming is putting together factsheets! Check out these new resources on attracting native bees and building pollinator habitat. You can also find the links on our ‘Resources Page’.
Spring is on its way and plenty of opportunities to attract our native pollinators are coming with it!
1 Comment
The last cattle are off the last fall grazed field. All the samples are all collected. The freezers are full of more insects, forage, and soil samples than you could ever imagine. So, what have our researchers, technicians, graduate students, and intrepid undergraduates been up to all this time? Soils TeamThe soils team has been hard at work processing fertility samples collected from the spring planted plots. An important measure of soil health is wet aggregate stability, which allows us to assess the resilience of the soil aggregates when disrupted by water. A soil sample down to 5 centimeters was collected and gently broken up into small pieces to fit through an 8 millimeter (about 1/3 inch) sieve spaces. After the soil dries, we put it on a sieve with smaller holes (2 millimeters), submerge the soil on the sieve for a few minutes, and then gently lift in the soil on the sieve in and out of the water for 2 minutes. The large stable aggregates will stay on top of the sieve, and the rest of the soil will pass through. The soil that passed through then goes on to a smaller mesh sieve (250 micron mesh size), and the process is repeated. The soil that passes through that sieve is then poured on top of an even finer mesh sieve (53 micron), and submerged repeatedly, as before. The soil on top of each sieve is dried and weighed to get our final data, which tell what percent of the soil is large or small aggregates. Pollinator TEAMCollections of wild bees were performed between late May and early September using blue vane traps. Since then the pollination biology lab has begun the preliminary steps of specimen identification by pinning and separating specimens by morphological characters. Once processed, each bee is identified down to genus (although species information is recorded where possible) and a tally is taken to map the diversity and abundance of bee pollinators within the field for each treatment (i.e. fallow, grazed, or ungrazed). We were lucky enough to recover several thousand bee specimens over the course of the summer and are looking forward to reporting our findings. Forage TEAMCover crops were clipped before, during, and after cattle grazing. Cover crops and weeds are then sorted by species. By sorting the cover crops, we can gain a better understanding of what species grow well on the High Plains and what was available for the cattle to graze on. Sorting individual weed species helps us understand the impact grazing has on the weed populations. While sorting doesn’t sound like the most challenging activity, it takes a lot of time and patience. Most of the weed species we’ve encountered are common and easy to identify, but every now and then, a species comes along that we haven’t seen yet. It’s always a lot of fun to identify a plant you’ve never seen before! After clippings are sorted, they are dried, weighed, ground, and finally forage quality is assessed using NIR. Sorting of the 2017 spring planted cover crops is almost done and we can look forward to compiling that data. Soil season has arrived! This past month was spent collecting soil samples from both the spring-planted and post-wheat cohorts for analysis. Fields that were finishing up with spring grazing were sampled down to 6 feet for moisture, and will be analyzed for other soil health and fertility indicators including soil carbon and nitrogen, bulk density and aggregation. Check out the video file below to see the hydraulic probe in action.
Fields that are preparing to plant the cover crop mix for the fall (or have already planted) were sampled for baseline soil moisture and fertility.
Wow, it has been a busy few weeks! Manhart, Thompson, and Sayles finished grazing this week, and wheat harvest is in full swing. We are getting ready for the post wheat planting of the cover crop and preparing experimental designs for the fields. It's been a while since we made a post, so I've accumulated a lot of great photos and videos. We've also gotten a lot of great photos/videos from producers. Keep them coming! We really enjoy getting to see what things look like when we're not able to be there. My personal favorite is the video from Feikert showing the monitor during wheat harvest. It's so exciting!!! We hit the road again this week for Kansas. On the docket this week was another set of weed transects, forage sampling, and cattle weights. While cattle were getting their measurements taken, our crew hit the field to count some weeds. Why count the weeds? Besides forcing undergraduates to learn weed identification, knowing the weed population at the start of the season will help us evaluate the weed suppressive abilities of both the cover crop and grazing. It will also help us understand the weed populations over the seasons and years. We’re planning on doing these transects when the cattle go on the field and after the cattle come off. Transects will be repeated in the following wheat crop to evaluate the weed control resulting from our efforts. That’s the management goal: control weeds now so there are less next year. Speaking of weeds, once we clip forage samples in the field, we bring them all back to the lab at CSU and sort each by plant species. This part is just to make another undergraduate learn their plant id (there’s a scientific reason too, I promise). Look forward to us to (and our fuzzy helpers) counting away in the field all summer! We found some very odd weeds Purple top turnip after the foliage has been clipped off.
The weather is warm and the cover crop is growing fast. The spring mix contains a nice mix of oats, barley, triticale, peas, flax, safflower, sunflower, rapeseed, and purple top. We are currently weighing cattle as they go onto the field, taking forage samples, and investigating the weed community using transects. The forage samples will be taken back to the lab, sorted by species, and an oven-dried weight will be taken on all the different species represented in each sample.
Augustine Obour with Kansas State University organized a successful cover crop field day at the HB Ranch near Brownell, Kansas, on May 19th. The day was wet and cool, but many turned out to hear about KSU research looking at spring and summer cover crops that are grazed, hayed, or left in the field. We also presented preliminary results from Year 1 of the grazed cover crop on-farm study.
Treatments have been established for three out of the six fields provided to us by producers participating in the spring cover crop grazing. The wheat near Bucklin was at heading or getting close. In Almena and Grainfield the wheat was still in the boot, but getting close! There were a lot of beautiful sites on this journey. I've included a few neat pictures at the end.  Now that everyone has planted and the cover crop is officially growing, it's time to lay out the experiments! The plan was to lay out the experiments three weeks after planting, but some of the fields have already been planted for a month or more. Since things tend to green up faster the farther east one goes, we decided to start with the eastern locations and work our way west. I will be in contact with the producers on the project shortly to let them know when to expect us. Thanks All! We've had a great start to this season. I've enjoyed the opportunity to visit everyone’s fields for spring soil moisture sampling, and I'm excited to say that everyone has planted. It was a dry start to the season but luckily, we have had a little moisture move through to help get things started. I will be posting updates, plans, and pictures here regularly. In addition, I will be sending out shorter updates via email. Please feel free to share or post comments and updates as well. Now that we have made it through the first round of soil moisture sampling, it is time to lay out the treatments! I would like to say a special thank you to John and Wilma. I always learn so much from the two of you. Thank you for all of your help! Lance Feikert
Larry and Doug Manhart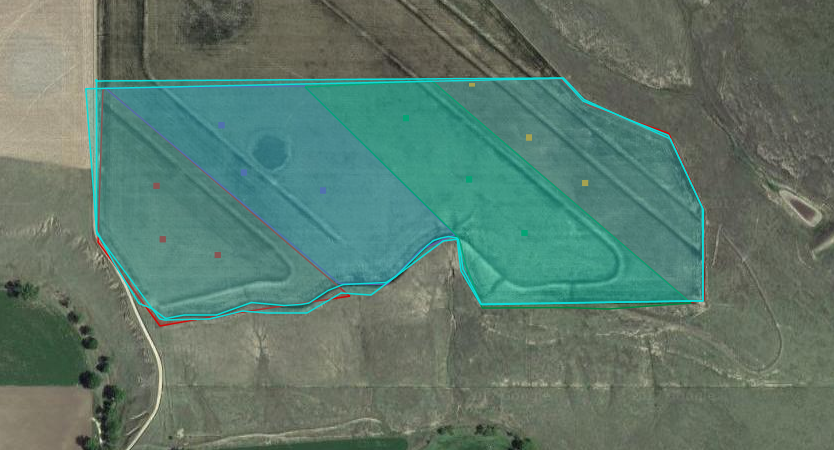 Wilma and I went to Larry and Doug Manhart’s March 10, 2017. It was cold, but sampling went well. The soil and soil moisture appeared to be highly variable across the 100-acres we sampled. There were spots where it was so dry that getting a sample was easy and others where it was too wet to take a sample. The field slopes and has many terraces. We arranged the blocks in the field to go with the terraces and tried our best to sample between them. The field was planted six days later on March 16, 2017. Curtis SaylesTodd Farnik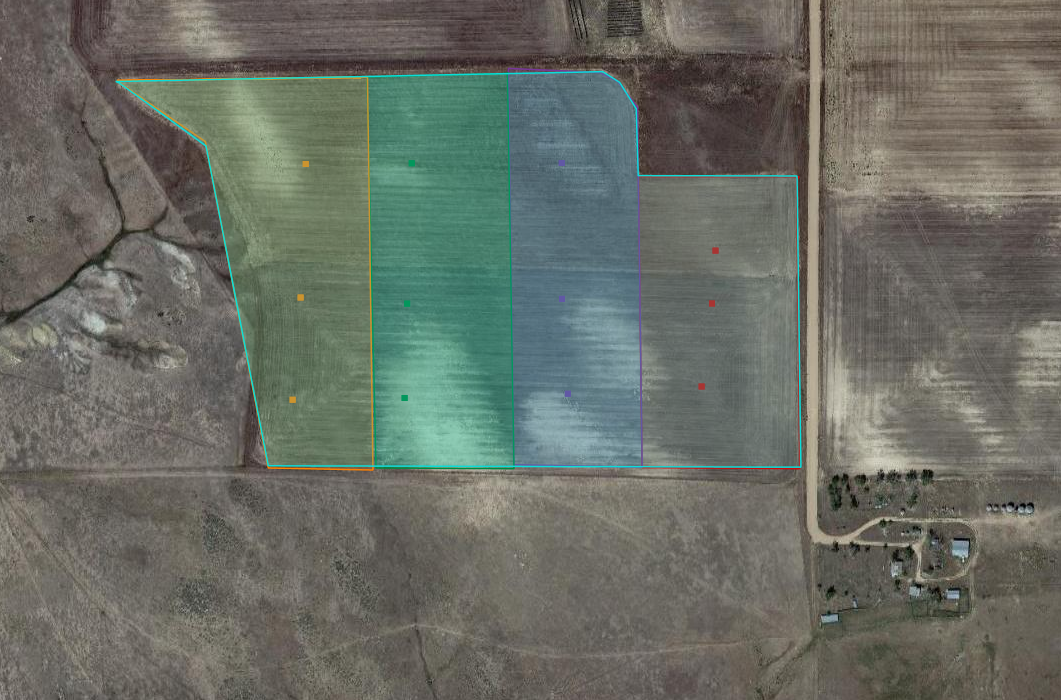 Our next adventure to Todd Farnik’s field was a bit more challenging. Again, we got lucky, because Todd was planting his field when we pulled up to sample. The field is 43-acres and was planted on March 23, 2017. Our challenge that day was the layer of sand and/or bedrock under the field that varies in depth. We also had problems with the probe and the GPS system, but we got the job done. Thankfully, our longest day of work had the shortest drive home. Michael ThompsonSteve Tucker |
Details
AuthorsThis blog is maintained by researchers at Colorado State University Archives
February 2018
Categories |
|||||||||||||||||||||||


















































 RSS Feed
RSS Feed
